HT1080-hNIS-Neo/iRFP-Puro
| Species | Human |
| Cell Type | Fibrosarcoma |
| Transgenes | Human sodium iodide symporter (hNIS) Near-infrared fluorescent protein (iRFP; ex/em 690/713nm) |
| Selection Genes | Neomycin (Neo) Puromycin resistance (Puro) |
-
Description
HT1080-hNIS-Neo/iRFP-Puro is a polyclonal population of the human fibrosarcoma cell line HT1080 (ATCC® CCL-121™). To achieve stable reporter expression in the polyclonal population, parental HT1080 cells were transduced with LV-hNIS-Neo (LV013) and LV-iRFP-P2A-Puro (LV032) and selected using G418 and puromycin. LV-hNIS-Neo encodes the human sodium iodide symporter (hNIS) cDNA linked to the neomycin resistance gene (Neo) via an internal ribosomal entry site (IRES) under the spleen focus-forming virus (SFFV) promoter. LV-iRFP-P2A-Puro encodes the near-infrared fluorescent protein (iRFP) cDNA linked to the puromycin resistance gene (Puro) via a P2A cleavage peptide (P2A) under the SFFV promoter.
*The ATCC trademark and trade name and any and all ATCC catalog numbers are trademarks of the American Type Culture Collection.
This cell line has been tested for mycoplasma contamination and is certified mycoplasma free.
The parental HT1080 cell line has been authenticated and certified free of interspecies cross contamination by short tandem repeat (STR) profiling with 9 STR loci.
-
Characterization
Morphology
 Low and high density cell morphology (200x)
Low and high density cell morphology (200x)NIS Expression
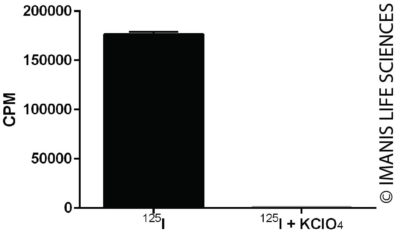 Cells were incubated with I-125 for one hour in the presence or absence of KClO4, an inhibitor of NIS mediated iodine uptake. Radioiodine concentrated within the cells was measured with a gamma counter.
Cells were incubated with I-125 for one hour in the presence or absence of KClO4, an inhibitor of NIS mediated iodine uptake. Radioiodine concentrated within the cells was measured with a gamma counter.iRFP Expression
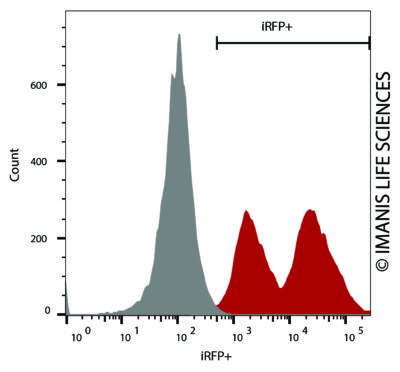 HT1080-hNIS-Neo/iRFP-Puro (red) or control (HT1080-Fluc-Puro) cells were fixed with paraformaldehyde and analyzed by flow cytometry (20,000 events).
HT1080-hNIS-Neo/iRFP-Puro (red) or control (HT1080-Fluc-Puro) cells were fixed with paraformaldehyde and analyzed by flow cytometry (20,000 events). -
Growth Conditions
Complete Growth Medium: DMEM supplemented with 10% FBS, 1X Penicillin/Streptomycin, 1.25 mg/mL G418, and 1 µg/mL puromycin.
The addition of G418 and puromycin to the complete growth medium maintains high dual reporter expression over continued passage of the cells. It is highly recommended, especially if the cells undergo multiple passages prior to being used for studies.
These cells should be grown in the indicated medium and passaged when they reach confluency. For routine passaging, cells are recommended to be split at a 1:10 ratio every 3-4 days.
-
Usage Information
These cells are suitable for in vitro and in vivo experimentation. HT1080 cells are a xenograph model for fibrosarcoma and form primary tumors and distant metastases (lung, liver, and brain) post implantation into immunosuppressed mice.1,2
The iRFP and NIS transgenes facilitate noninvasive fluorescence imaging and high-resolution 3D SPECT/PET imaging, respectively, of implanted cells. To reduce background autofluorescence, mice should be fed an alfalfa-free diet for at least a week prior to imaging.
The cells can be amplified in vitro and used to generate additional frozen stocks. Cryopreservation of low passage stocks is recommended. Frozen stocks should be preserved in a designated cryopreservation medium.
These cells were generated via lentiviral vector transduction. The lentiviral vector used for transduction was a self-inactivating (SIN) vector in which the viral enhancer and promoter have been deleted. Transcription inactivation of the LTR in the SIN provirus increases biosafety by preventing mobilization by replication competent viruses and enables regulated expression of the genes from the internal promoters without cis-acting effects of the LTR3. Nevertheless, all work with these cells should be performed under biosafety-level 2 (BSL2) conditions by trained personnel. Institutional requirements may permit handling of these cells under BSL1 conditions if certain criteria are met.
References:
1Rasheed et al. Cancer. 1974. 33:1027-1033.
2Praus et al. Gene Therapy. 1999. 6:227-236.
3Miyoshi et al. J Virol 1998. 72:8150-8157. -
Datasheet/COA
Lot Number CL-IM105