B16F10-Fluc-hNIS
| Species | Mouse |
| Cell Type | Melanoma |
| Transgene | Firefly Luciferase (Fluc) Human Sodium Iodide Symporter (hNIS) |
-
Description
This is a population derived from the melanoma B16F10 cell line (ATCC® CRL-6475TM). Parental B16F10 cells were transduced with LV-Luc2-P2A-hNIS (Imanis #LV023) encoding the firefly luciferase (Luc2; Fluc) cDNA under the spleen focus-forming virus (SFFV) promoter and linked to the human sodium iodide symporter (hNIS) cDNA via a P2A cleavage peptide. High Fluc and hNIS expressing cells were selected using a methylcellulose-based semi solid medium. The Fluc transgene facilitates noninvasive bioluminescent imaging.
*The ATCC trademark and trade name and any and all ATCC catalog numbers are trademarks of the American Type Culture Collection.
This cell line has been tested for mycoplasma contamination and is certified mycoplasma free.
The parental B16F10 cell line has been authenticated and certified free of interspecies cross contamination by short tandem repeat (STR) profiling with 9 STR loci.
Due to the immunogenicity of the reporter genes in this cell line, we recommend using immunocompromised mice for in vivo studies.
Replication Competent Lentivirus (RCL) Test (including a test report) is available for this cell line at an added cost. Contact us to learn more.
-
Reporter Validation
Morphology
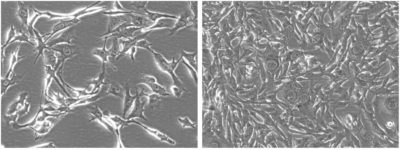
Low and high density morphology (200x)
Luciferase Expression
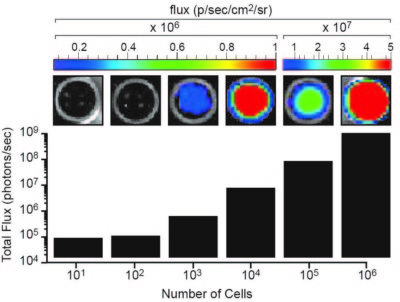
The indicated number of cells were placed in wells of a 96-well plate. After the addition of 3 mg/mL d-luciferin, the plate was immediately imaged using an IVIS Spectrum. The total flux (photons/sec) was plotted as a function of cell number.
NIS Expression
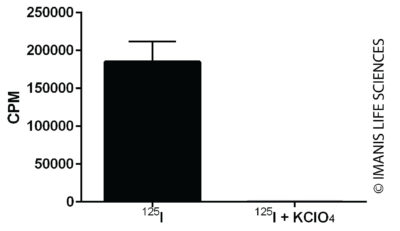 Uptake of I-125 by 3 x 105 cells was assayed in the presence or absence of KClO4, an inhibitor of NIS-mediated I -125 uptake.
Uptake of I-125 by 3 x 105 cells was assayed in the presence or absence of KClO4, an inhibitor of NIS-mediated I -125 uptake.In Vivo Imaging
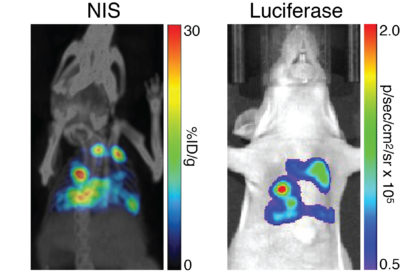 Nude mice were implanted intravenously with B16F10-Fluc-hNIS cells and tumor metastasis was monitored non invasively. Comparative images were acquired on day 23 by SPECT/CT imaging with [99mTc]-pertechnetate (left) of bioluminescent imaging with d-luciferin (right).”
Nude mice were implanted intravenously with B16F10-Fluc-hNIS cells and tumor metastasis was monitored non invasively. Comparative images were acquired on day 23 by SPECT/CT imaging with [99mTc]-pertechnetate (left) of bioluminescent imaging with d-luciferin (right).” -
Growth Conditions
Complete Growth Medium: Dulbecco’s Modified Eagle’s Medium (DMEM) supplemented with 10% FBS, 1X Penicillin/Streptomycin.
These cells should be grown in the indicated medium and passaged when they reach confluency. For routine passaging, cells are recommended to be split at a 1:10 ratio every 3-4 days.
B16F10 cells produce melanin; accumulation of melanin turns the cells dark brown or black and prolonged melanin secretion turns cell culture medium black. Melanin is toxic and B16F10 cells will die in the presence of excess melanin. Culture medium should be changed as soon as it becomes black, even if the cells are not confluent. Typically, media changes between passages are not required. Different lots of FBS have been observed to affect the rate of melanin production by B16F10 cells.
B16F10 cells trypsinize relatively quickly. Excess trypsinization can damage the cells and care should be taken to quickly neutralize the trypsin upon cell detachment.
-
Usage Information
These cells are suitable for in vitro and in vivo experimentation. B16F10 cells form tumors and pulomary metastases post implantation into syngenic C57BL/6 mice.1 The NIS transgene facilitates high resolution, 3D SPECT/PET imaging of implanted cells. NIS is not immunogenic and is therefore suitable for longitudinal imaging studies in a syngenic model.
The cells can be amplified in vitro and used to generate additional frozen stocks. Cryopreservation of low passage stocks is recommended. Frozen stocks should be preserved in a designated cryopreservation medium.
These cells were generated via lentiviral vector transduction. The lentiviral vector used for transduction was a self-inactivating (SIN) vector in which the viral enhancer and promoter have been deleted. Transcription inactivation of the LTR in the SIN provirus increases biosafety by preventing mobilization by replication competent viruses and enables regulated expression of the genes from the internal promoters without cis-acting effects of the LTR2. Nevertheless, all work with these cells should be performed under biosafety-level 2 (BSL2) conditions by trained personnel. Institutional requirements may permit handling of these cells under BSL1 conditions if certain criteria are met.
References:
1Fidler. Cancer Res. 1975. 35:218-224.
2Miyoshi et al. J Virol 1998. 72:8150-8157. -
Datasheet/COA
Lot Number CL-IM153